Young people in Leeds being invited to take part in national Covid booster jab trial
and live on Freeview channel 276
Leeds Teaching Hospitals NHS Trust is inviting people aged 18-30 to take part in the COV-BOOST: Young Adults Fractional Dosing Sub-study and volunteers will be paid for their time.
The study will determine whether a lower dose of the Pfizer or Moderna booster vaccines give as good an immune response in young adults as higher doses.
Advertisement
Hide AdAdvertisement
Hide AdLower doses may also be linked with fewer side effects or lower rates of already rare adverse events, researchers believe.
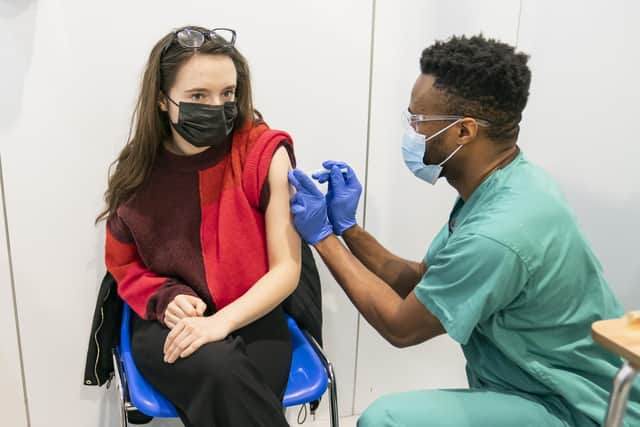

Here's everything you need to know about the trial:
Why is the study taking place?
Professor Saul Faust, Chief Investigator and Director of NIHR Southampton Clinical Research Facility, said: "The first phase of the COV-BOOST study looked at the safety, immune responses and side-effects of seven COVID-19 vaccines when used as a third booster jab.
"The findings have helped shape the UK booster programme and given important evidence towards global vaccination efforts.
“In this next phase, we are looking for volunteers aged 18 to 30 to help us investigate the safety and side effect profile of giving lower doses of either the Pfizer or Moderna COVID-19 vaccines.
Advertisement
Hide AdAdvertisement
Hide Ad“If we find that giving a lower dose of these vaccines to young adults gives as good an immune response as a higher dose, this could have positive implications for global vaccine supply and may result in a lower side effect profile in this age group. "
Who can take part in the trial?
Dr Phil Wood, Chief Medical Officer at Leeds Teaching Hospitals NHS Trust, said: Throughout the pandemic, the participation of people from across Leeds and West Yorkshire in vaccine studies has been critical in improving our understanding of vaccine-related protection from COVID-19.
"To support on-going research into COVID-19 vaccinations, the Trust is inviting people aged between 18 to 30 years old to be part of research.”
The NIHR-supported study is looking for volunteers who are:
- are between 18 to 30 years old
- have had two doses of the Pfizer or Moderna COVID-19 vaccines, with at least 3 months (84 days) since their second dose
- have not received a booster
Advertisement
Hide AdAdvertisement
Hide AdPeople who have tested positive for COVID-19 in the past, and have had their second vaccine, can also take part in the study.
What will happen during the clinic?
Participants will be randomly selected to receive one of the following doses:
- A single dose of Pfizer (currently used in the UK booster programme)
- One third of a single Pfizer dose (currently recommended for 5 - 11 years old in the UK)
Advertisement
Hide AdAdvertisement
Hide Ad- A half Moderna dose (the dosage used by the NHS as a 3rd dose booster)
- One quarter of a single Moderna dose
When and where will the trial take place?
The booster jabs will be administered at clinics planned for January 27 and 28 at St James Hospital’s Bexley Wing.
The clinics will run between 9am and 2:30pm.
Will I get paid for the trial?
Yes - people who take part will be reimbursed up to £225 for their time, inconvenience and travel.
How many volunteers will take part in the trial?
Around 900 volunteers will be recruited from 15 research sites across the country, as well as in Leeds.
The other sites include:
- University of Hospitals of Leicester NHS Trust
- University College London Hospitals
- Guys' and St Thomas' NHS Foundation Trust
- St George's University Hospital
- London North West University Healthcare NHS Trust
- University Hospital Southampton NHS Foundation Trust,
- Portsmouth Hospitals University NHS Trust
- University Hospitals Dorset NHS Foundation Trust
- Public Health Wales
- Wrexham Maelor Hospital
- Cambridge University Hospitals
Advertisement
Hide AdAdvertisement
Hide AdFor further information regarding the study, contact the Covid-19 research team at Leeds Teaching Hospitals NHS Trust via [email protected] or 07425 628243.
Support the YEP and become a subscriber today. Enjoy unlimited access to local news and the latest on Leeds United. With a digital subscription, you'll see fewer ads, enjoy faster load times, and get access to exclusive newsletters and content. Click here to subscribe